Science’s COVID-19 reporting is supported by the Pulitzer Center and the Heising-Simons Foundation.
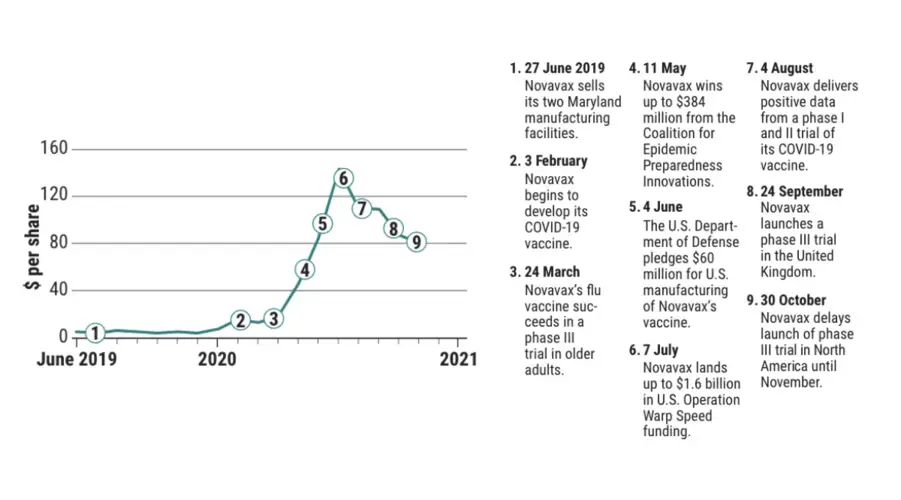
GAITHERSBURG, MARYLAND—Eighteen months ago, a small vaccinemaker here called Novavax faced an existential threat: delisting by the NASDAQ stock index. On the heels of a second failed vaccine trial in less than 3 years, the firm’s shares had plunged to less than $1 for 30 straight days, triggering a warning by NASDAQ. Frantic to conserve cash, the company sold its two Maryland manufacturing facilities, slicing its payroll by more than 100 employees. By January, it employed only 166 people.
“Good ideas. Bad management. … The company will probably die soon,” a former Novavax manager wrote on Glassdoor.com in October 2019.
What a difference a year—and a pandemic—make. Today, Novavax is slated to receive up to $2 billion from the U.S. government and a nonprofit organization to develop and manufacture a coronavirus vaccine. The company’s stock closed at $80.71 per share on 30 October, it has hired more than 300 new employees, and this month it plans to launch a pivotal clinical trial of its coronavirus vaccine in the United States and Mexico. Made by moth cells harnessed to crank out the virus’ spike protein—which the pathogen uses to invade human cells—Novavax’s vaccine outshone major competitors on key measures in monkey and early human tests.
The company is one of just seven vaccinemakers to win funding so far from Operation Warp Speed, the giant multiagency U.S. government effort aiming to quickly produce at least 300 million doses of COVID-19 vaccines. But most Warp Speed–backed companies are giant pharmaceutical firms, and most have already launched late-stage clinical trials in the United States. Tiny Novavax is rushing to keep pace with its larger rivals because companies that win the first approvals from regulators will have big market advantages. Still, some observers say Novavax’s technology gives it an edge.
“They are incredibly well positioned,” says Andrew Ward, a structural biologist at Scripps Research. Ward, who receives no payments from the company but owns some stock, led a team that last month published a paper in Science describing the structure of Novavax’s tailormade spike protein, the heart of its vaccine. He was impressed by its stability and conformation, as well as the vigorous antibody responses it has elicited in humans and animals. “They have the know-how,” he says. “And they obviously, as we confirmed, make a good product.”
But other people are skeptical. They note that Novavax has focused on making vaccines for more than 20 years but has never brought one to market, and that its senior executives have sold tens of millions of dollars of company stock since its share price began to soar this summer.
Most significantly, the company has an Achilles’ heel. Novavax must rely mostly on contract manufacturers to meet its ambitious goal for 2021: producing enough vaccine to give 1 billion people two shots each. If manufacturing problems crop up—and the company last week said manufacturing delays had slowed launch of its late-stage North American trial—competing vaccines may surge ahead. “That’s concerning,” says David Maris, a veteran drug industry analyst and managing director at Phalanx Investment Partners. Where small companies such as Novavax are concerned, he adds, “people do want to believe in fairy tales.”
ON 10 JANUARY, researchers in China published the genome sequence of the virus ravaging the city of Wuhan. Three days later, Gregory Glenn, president of R&D at Novavax, asked his staff to order from a supplier the gene for the virus’ spike protein.
Glenn and other Novavax scientists had spent years developing “protein subunit” vaccines, so named because they employ a protein (or part of one) from the targeted virus, plus an immune-boosting compound called an adjuvant, to provoke an immune response. The company hadn’t had a commercial success—its vaccine against a serious respiratory illness failed in clinical trials. But it had produced a promising flu vaccine aimed at older adults, which was nearing the end of a pivotal trial. The company had also created protein subunit vaccines against two close cousins of the pandemic virus—the coronaviruses that cause severe acute respiratory syndrome and Middle East respiratory syndrome, using those viruses’ spike proteins. Those vaccines hadn’t made it to market, but Novavax had plenty of experience with the coronavirus family. Glenn believed it was his company’s moment.
The gene for the spike protein was slow to arrive, however. Finally, at 6 a.m. on 3 February, a vice president from the supplier hand-delivered a red-capped vial bearing the gene to Novavax’s beige brick building here. The virus still hadn’t been officially named—the vial was labeled “Cov/Wuhan”—but Novavax was now out of the gate and in the race to tame it.
The company’s scientists started to work “with frenetic pace,” Glenn says. Some of their competitors were already a lap ahead, working on their own vaccines. “There’s no question [that we’re] behind” several companies that also won Warp Speed funds, Glenn said on the morning of 24 September, the day Novavax launched its first phase III trial, of 15,000 volunteers in the United Kingdom.
Most of Novavax’s key competitors—Moderna, Pfizer, Johnson & Johnson subsidiary Janssen, and AstraZeneca—had launched phase III trials by then. To make their vaccines, all four of those firms use new technologies based on genetic material that directs protein production, rather than delivering proteins directly. Those platforms rely on DNA loaded in disabled viruses or on messenger RNA to carry genetic instructions for building the spike protein. Cells within a vaccinated person then churn out the protein, alerting the immune system.
Developers of protein vaccines must develop their own version of the spike protein—one that closely mimics the naturally occurring spike and is stable enough to retain its immunological punch during manufacturing, packaging, and distribution. Most such vaccines include an additional compound called an adjuvant to help stimulate a strong, protective immune response. Those extra steps make protein vaccines slower to develop than those that deliver genetic instructions.
But protein-based vaccines also have a long track record of effectiveness, in contrast with the newer, largely unproven approaches. The successful hepatitis B vaccine licensed in 1986 and recommended for all U.S. babies in their first day of life is a protein subunit vaccine. So are a flu vaccine approved in 2013 and the human papillomavirus vaccines that have sent rates of cervical cancer plunging since the first ones were licensed in the 2000s.
Perhaps because the technology is tried and true, scores of other companies are also racing to develop protein subunit vaccines. Novavax is the only one to have launched a phase III trial. Of the other firms, the huge vaccinemaker Sanofi Pasteur is likely Novavax’s biggest rival. It “is going to be formidable competition to the Novavax vaccine,” says Vijay Samant, a former head of vaccine manufacturing at Merck and now a consultant to vaccine companies. (Novavax is not a client.) Sanofi Pasteur has deep pockets, infrastructure, and experience, and markets vaccines against 19 infectious diseases.
But Novavax scientists say they’re ready for the competition. “We’ve been getting ready for this our whole lives,” says Gale Smith, Novavax’s chief scientist.
ONCE THE PANDEMIC coronavirus gene arrived in Gaithersburg, Maryland, on 3 February, the company spent weeks making more than 20 versions of the spike protein, aiming for a product as immunologically potent as possible. The winner was the most stable antibody-inducing protein, one that mimicked the energy-packed state of the spike just before it fuses with the host cell membrane.
In March, a team led by Nita Patel, a senior director in the vaccine development department (see sidebar), confirmed in lab tests that the engineered protein bound tightly to its human cell-surface receptor. The results strongly suggested antibodies to Novavax’s protein would interfere with the virus’ own spike protein as it tried to fuse with cells.
Patel’s boss, Smith, next enlisted Ward to verify the protein’s structure and stability with electron microscopy. Other tests showed the Novavax spike is stable for many weeks at 2°C to 8°C—a key advantage over the Moderna and Pfizer vaccines, which need to be stored at –20°C and –70°C, respectively, and once thawed, last only days in the refrigerator.
Now, the challenge was to make the protein in the vast quantities that the world would need. Novavax had a system to do that, co-invented by Smith decades earlier and since used by the company to develop its other vaccine candidates: moth cells.
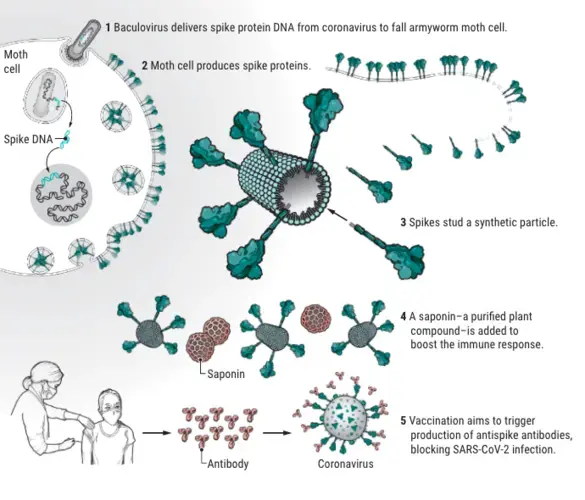
As a 34-year-old graduate student at Texas A&M University in 1983, Smith, with colleagues, had developed a system that could produce proteins in big quantities. The researchers started with an insect-infecting virus called a baculovirus, which had the virtue of a roomy genome that can accommodate large chunks of foreign DNA. The researchers inserted a gene for a human immune protein, interferon, into the virus and then used it to infect cells from the caterpillar form of a pest called the fall armyworm moth. The virus transferred the gene to the moth cells, which duly secreted human interferon.
Back then, editors of major journals had little interest in the discovery and repeatedly rejected Smith’s paper, which found a home in an obscure new journal, Molecular and Cellular Biology. But today the system is widely used in biotechnology. Now, it is at work producing the Novavax vaccine at a plant owned by a contractor in Morrisville, North Carolina, and soon, it’s expected, at other plants owned or contracted by Novavax in Europe, the United States, and Asia.
In North Carolina, countless baculoviruses loaded with the gene for the coronavirus spike protein are invading moth cells in 2000-liter bioreactors. The moth cells then express the coronavirus spikes on their cell membranes. Scientists harvest the proteins and mix them with a delivery vehicle: synthetic particles, on average 30 to 40 nanometers across. Each “nanoparticle” ends up studded with up to 14 spike proteins. The finished particles are only slightly smaller than the coronavirus itself, helping the immune system “see” them as a danger, Smith says. Then Novavax adds its adjuvant, based on saponin, a compound found in soap bark trees that stimulates the immune system (see graphic, above).
To crank up production in the spring, Novavax had to cope with a harsh reality. “What we didn’t have—what we’d lost by downsizing last year—is taking [the vaccine] right over to a manufacturing facility,” Glenn says. “We could have been much earlier had we had that strategic asset.” Instead, the company “had to beg,” he says, turning to Gaithersburg-based contract manufacturer Emergent BioSolutions to produce vaccine for the first human trials. It was “not ideal,” Glenn says. “We’re sprinters … [but] it’s hard to get someone else to sprint.”
A vital piece of good news came on 24 March, boosting the company’s search for cash. The phase III clinical trial of Novavax’s flu vaccine in older adults, also made using the moth cell system, returned excellent results. For Novavax, which was now asking funders for major dollars for its pandemic coronavirus vaccine, the proof of its capabilities came at a fortuitous time.
Three days later, senior Novavax scientists met with decision-makers from a key funder—the nonprofit, international Coalition for Epidemic Preparedness Innovations (CEPI)—in a crucial Zoom call. Novavax scientists were used to dealing with skeptics. “Most people don’t think [our nanoparticles] work,” Smith says. “We’ve been accused that [they] aren’t even real, which is ridiculous.” Then Ward’s new high-resolution images—showing tiny particles studded with spike proteins, with honeycomblike saponins floating beside them—flashed on dozens of people’s screens. It was “one of those precious moments,” recalls Nick Jackson, CEPI’s head of vaccine programs. “The excitement was palpable.”
Six weeks later, CEPI awarded Novavax up to $384 million for trials and manufacturing of its vaccine.
Ward’s work won over some scientific doubters. John Moore, an immunologist at Weill Cornell Medicine, had been skeptical of Novavax’s moth cell system because in the 1990s it had conspicuously failed to produce an HIV spike protein with the right characteristics to make an AIDS vaccine. But in August, when Ward’s work was posted as a preprint, “I looked at that paper and was impressed,” Moore says. “It changed my perception of the quality of the protein. The concerns I had were eliminated by data, which is as it should be.”
By late May, Novavax had launched its first human safety trial in 131 volunteers in Australia and used the CEPI funding to buy, for $167 million in cash, a state-of-the-art vaccine manufacturing facility in the Czech Republic that the company said would deliver more than 1 billion doses in 2021. And in early July, Operation Warp Speed granted the company up to $1.6 billion, with $800 million available immediately, for a phase III clinical trial and for manufacturing 100 million doses of vaccine.
IN EARLY AUGUST, the big investors in the tiny company won an initial vindication when Novavax announced strong results from the Australian trial. After two injections, “the antibody responses in the Novavax paper were markedly stronger than any of the other vaccines that have been reported,” and participants had experienced no severe adverse events, says Moore, who recently published a Journal of Virology review of the leading vaccine candidates. Moore says he intends to volunteer for a Novavax trial if eligible. “I’m going, ‘Yes, I’ll have that [vaccine].’”
The government of the United Kingdom soon signed up to buy 60 million doses of Novavax’s vaccine, and the big drugmaker Takeda licensed it to manufacture at scale with funding from the Japanese government. Other scientists noted strong results in a dozen monkeys injected with various doses of Novavax’s vaccine and then infected with live coronavirus. The virus failed entirely to multiply in the animals’ noses and replicated in the lungs of just one monkey that received the lowest dose; that animal shut down the infection after 4 days.
“It’s the only vaccine I’ve seen out of all the candidates that are further down the pipeline that actually had no viral replication in the nasal swabs of vaccinated animals,” says Angela Rasmussen, a virologist at Columbia University. That’s important, she says, because stopping viral replication in the nose can reduce the spread of infection among people who may be unaware they are sick. But she cautions that monkeys are not people. “We can’t really conclude that this vaccine is going to be better in practice until we have some reliable safety and efficacy data in people.”
That’s why researchers will be eager to see results from Novavax’s phase III trials. In the one in progress in the United Kingdom, volunteers get either placebo or vaccine in two injections, 21 days apart. Researchers will evaluate whether vaccinated volunteers have fewer symptomatic coronavirus infections than placebo recipients. They’ll also keep monitoring safety, including any reactions to the adjuvant, because this will be the first time huge numbers of younger people with vigorous immune systems receive it.
Vaccine trials need volunteers who are exposed to the virus, so skyrocketing infections in the United Kingdom are likely working in Novavax’s favor. The company estimates there will be enough infections among participants to allow a first look at the data early in 2021—and says it expects the U.K. data will be enough to drive approval of its vaccine.
In the United States and Mexico, the company aims to enroll 30,000 volunteers—about 90% of them in the United States—in a trial expected to launch this month. Keeping volunteers in that trial may be challenging if other vaccine contenders win U.S. marketing approval in coming weeks. But Glenn argues that this scenario is “not very realistic” given the time required for U.S. Food and Drug Administration (FDA) action and widespread distribution of a vaccine.
Observers also note that more than one vaccine will probably be needed to quell the pandemic. The first round of vaccine doses promised by Pfizer and Moderna combined would only cover 100 million people. Experts add that the first vaccine approved won’t necessarily be the best. “Especially when we are talking about vaccinating millions of people, you have just got to follow the science,” says Mayank Mamtani, a senior biotechnology analyst at B. Riley Securities. In the long run, “being first to market is just not, in my opinion, important.”
SOME OBSERVERS RAISED their eyebrows when Novavax won Warp Speed funding, given that the firm was on life support last year. Maris bemoans the “complete lack of transparency as to how the funding decisions of Warp Speed were made.” (Journalists and investors have tried unsuccessfully to obtain Novavax’s Warp Speed contract.) “I would like to see the fundamental or deep analysis, if any, done prior to these funding decisions,” Maris says. “I’m not certain whether it exists.”
Novavax says it will release the contract next week as part of its quarterly financial reporting.
Other critics noted that since the pandemic began, Novavax top managers have made tens of millions of dollars by exercising options to buy their company’s stock at earlier, lower prices and sell it at this year’s high prices. Such options are a common way to pay biotech executives, but against the background of a pandemic, the sales led to an unwelcome spotlight on Capitol Hill in September. At a hearing, the chair of the House of Representatives Financial Services Subcommittee on Investor Protection, Entrepreneurship, and Capital Markets raised the examples of Novavax and other companies by name. And in a stinging memo that Democratic lawmakers issued under the heading “Corporate Integrity and Profiteering in the COVID-19 Pandemic,” they reported that three senior executives at Novavax and two board members had pocketed nearly $17 million in August alone.
Maris notes that executives who are bullish on their own companies tend to keep their stock. “So it’s interesting that they decided to sell.”
Novavax leaders continued their sales in September, with four senior managers selling shares worth a combined $18.9 million. “Pretty much all my value I have is in Novavax,” says Glenn, who pocketed $6.2 million (before taxes) in August and sold shares worth $4.9 million in September. “A year ago it was worth nothing.” CEO Stanley Erck, who sold $4.5 million in stock in September, did not respond to requests for comment.
Even if Novavax’s phase III trials succeed, vaccine watchers say the company’s biggest vulnerability will remain: the manufacturing process. “I think the vaccine is going to be efficacious,” Samant says. “But you put that 2 billion–dose task in front of a small company, it’s a huge uphill task.”
Ward concurs. “It’s like trying to cook a really good quiche. You can make one for your family, but if you try to make 50 of them for a wedding, that’s actually very difficult to do, to get them all the same and equivalent.”
“The [moth cell system] is not like the flu vaccine and eggs,” adds vaccine expert and FDA veteran Luciana Borio, an admirer of Novavax who is a vice president at In-Q-Tel, a technology investment firm. “It’s untested.”
Novavax counters that the company’s manufacturing partners are deeply experienced in making vaccines at scale. For instance, the Serum Institute of India, a veteran vaccinemaker that is the world’s largest, is helping Novavax expand production at the 14,000-square-meter Czech facility, plus manufacturing an additional billion doses itself, in India, for low- and middle-income countries. That brings the total doses the company now says it can manufacture in 2021 to 2 billion, the promised number.
The fact that world-class manufacturers such as the Serum Institute “didn’t even blink before wanting to work with them and helping them scale up” speaks highly of the company, Mamtani says.
“It’s going to be a dark horse,” he predicts. “A dark horse helping us to get out of this darkness.”
COVID-19 Update: The connection between local and global issues–the Pulitzer Center's long standing mantra–has, sadly, never been more evident. We are uniquely positioned to serve the journalists, news media organizations, schools, and universities we partner with by continuing to advance our core mission: enabling great journalism and education about underreported and systemic issues that resonate now–and continue to have relevance in times ahead. We believe that this is a moment for decisive action. Learn more about the steps we are taking.