Since the gene-edited babies known as Lulu and Nana became international news in November 2018, scientific debate and media speculation have swirled around the potential impacts of modifying their gene for CCR5. One recent study prompted the MIT Technology Review to suggest the twins have enhanced memories and learning abilities, leading to copycat stories worldwide that exercised less restraint. And in June, drawing on a population analysis of variants of the gene CCR5 published in Nature Medicine, headlines blared that the girls might have shortened lives. "This interpretation is not valid or responsible," Rasmus Nielsen, a geneticist at the University of California (UC), Berkeley, who led the work, countered in a tweet.
A major concern has been that He Jiankui's attempts to cripple CCR5, the gene for a protein on immune cells that HIV uses to infect the cells, also made "off-target" changes elsewhere in the girls' genomes. Those changes could cause cancer or other problems. He contends that the babies have no such off-target mutations, although some scientists are skeptical of the evidence offered so far.
People inherit two copies of CCR5, one from each parent. He chose the gene as a target because he knew that about 1% of Northern European populations are born with both copies missing 32 base pairs, resulting in a truncated protein that doesn't reach the cell surface. These people, known as CCR5Δ32 homozygotes, appear healthy and are highly resistant to HIV infection.
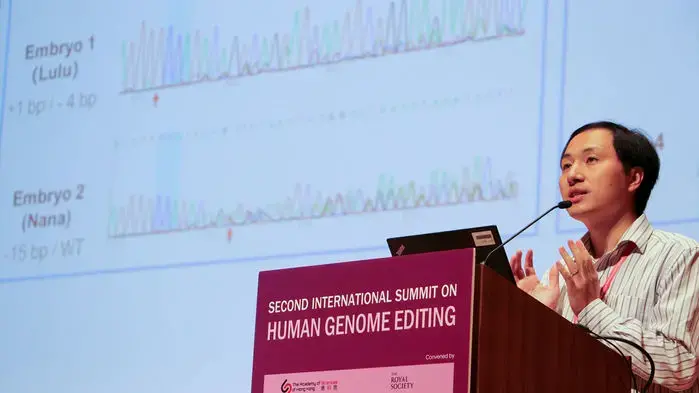
In the embryos He's team edited, the researchers did not attempt to delete these exact 32 base pairs; rather, the group designed CRISPR to cut CCR5 at the base pair at one end of the natural deletion. The error-prone cell-repair mechanism, which CRISPR depends on to finish knocking out genes, then deleted 15 base pairs in one of Lulu's copies of the gene, but none in the other. With one normal CCR5, she is expected to have no protection from HIV. Nana, according to the data He presented in a slide at an international genome-editing summit held in November 2018 in Hong Kong, China, had bases added to one CCR5 copy and deleted from the other, which likely would cripple both genes and provide HIV resistance.
He added the genes for the CRISPR machinery almost immediately after each embryo was created through in vitro fertilization, but several researchers who closely studied the slide caution that it may have done its editing after Nana's embryo was already past the one-cell stage. That means she could be a genetic "mosaic" who has some unaffected cells with normal CCR5—and ultimately might have no protection from HIV.
Immunologist Philip Murphy helped discover the role of CCR5 in HIV infection in 1996 and published early studies showing how the 32-base-pair mutation bestows HIV resistance. Murphy, of the National Institute of Allergy and Infectious Diseases in Bethesda, Maryland, cautions that He's CRISPR edits in either girl could cause harm by producing an altered protein with unpredictable functions. "This is a potential complication of editing that gets much less attention than potential off-target edits and effects," Murphy says.
The CCR5 protein naturally functions as a receptor for chemokines, which are immune system messengers, but the protein's exact impact remains mysterious. Murphy, in a mouse experiment he published in 2005, showed that CCR5 promotes trafficking of important immune cells to the brain during infection with West Nile virus. He later found that CCR5Δ32 homozygous humans who get infected with that virus are more susceptible to severe encephalitis and even death than other people. Some mouse and human studies have also found that CCR5Δ32 homozygotes get more severe symptoms from the influenza virus; however, other researchers dismiss this concern, pointing to conflicting epidemiologic analyses.
Evolutionary biologists have pondered why this mutation has survived in Northern European populations—it's virtually nonexistent in East Asia—and some have tied it to past survival advantages against scourges such as bubonic plague and smallpox. But those arguments have gained little traction. "All of the CCR5 disease associations with the exception of HIV/AIDS need substantially more experimental support before one could handicap risks of CCR5 editing with confidence," Murphy says.
In an attempt to better understand whether CCR5Δ32 homozygotes suffered any harm from the mutation, Nielsen's Nature Medicine study analyzes survival in some 400,000 people of U.K. ancestry who voluntarily provided their DNA for sequencing to the U.K. Biobank. The study found that CCR5Δ32 homozygotes were about 20% less likely than the rest of the population to reach age 76. "World's first gene-edited babies 'more likely' to die young," a headline in the United Kingdom's The Telegraph announced.
But people on average joined the study at 56.5 years old, so—despite the suggestions of the Telegraph report and many other news stories in reputable publications—the study says nothing about whether the mutation affects the survival of young people or those older than 76. In tweets, Nielsen stressed that his group also had "very large" error ranges because relatively few CCR5Δ32 homozygotes in the study had died so far. What's more, Lulu is clearly not a CCR5Δ32 homozygote, so the findings don't apply to her. As for Nana, she doesn't have the 32-base-pair mutation, and her Chinese genetic background means any changes to CCR5 might have a different impact, anyway.
The speculation about the twins' enhanced brain functions has even shakier support. Researchers showed in 2016 that knocking out one or both CCR5s in mice enhances their memory and cognition. A subsequent study that crippled CCR5 in mice found that, compared with control animals, the mutants recovered from strokes more quickly and had improved motor and cognitive functions following traumatic brain injury. The later study, in the 21 February issue of Cell, also included an analysis of 68 stroke patients who had one copy of CCR5 with the HIV resistance mutation; it concluded they had improved recovery, too. Extrapolating from this work, the MIT Technology Review headlined a story "China's CRISPR twins might have had their brains inadvertently enhanced."
Neurobiologist Alcino Silva of UC Los Angeles, a co-author of both mouse studies, says his team's findings "strongly suggest that the CCR5 manipulation changed cognitive function," but the researchers have seen both positive and negative effects. "It is unfortunate that there is the misperception that we know how to engineer 'smarter' babies," Silva says—a point the MIT Technology Review article does underscore. He concludes it is "way too early" to conduct experiments like the one He did. "We do not know enough, and the consequences can be disastrous," he says.
Or the editing of CCR5 in Lulu and Nana could have no detectable consequences at all. With no one providing updates on the health of the children, that outcome, perhaps the most likely one, may never be headline news.